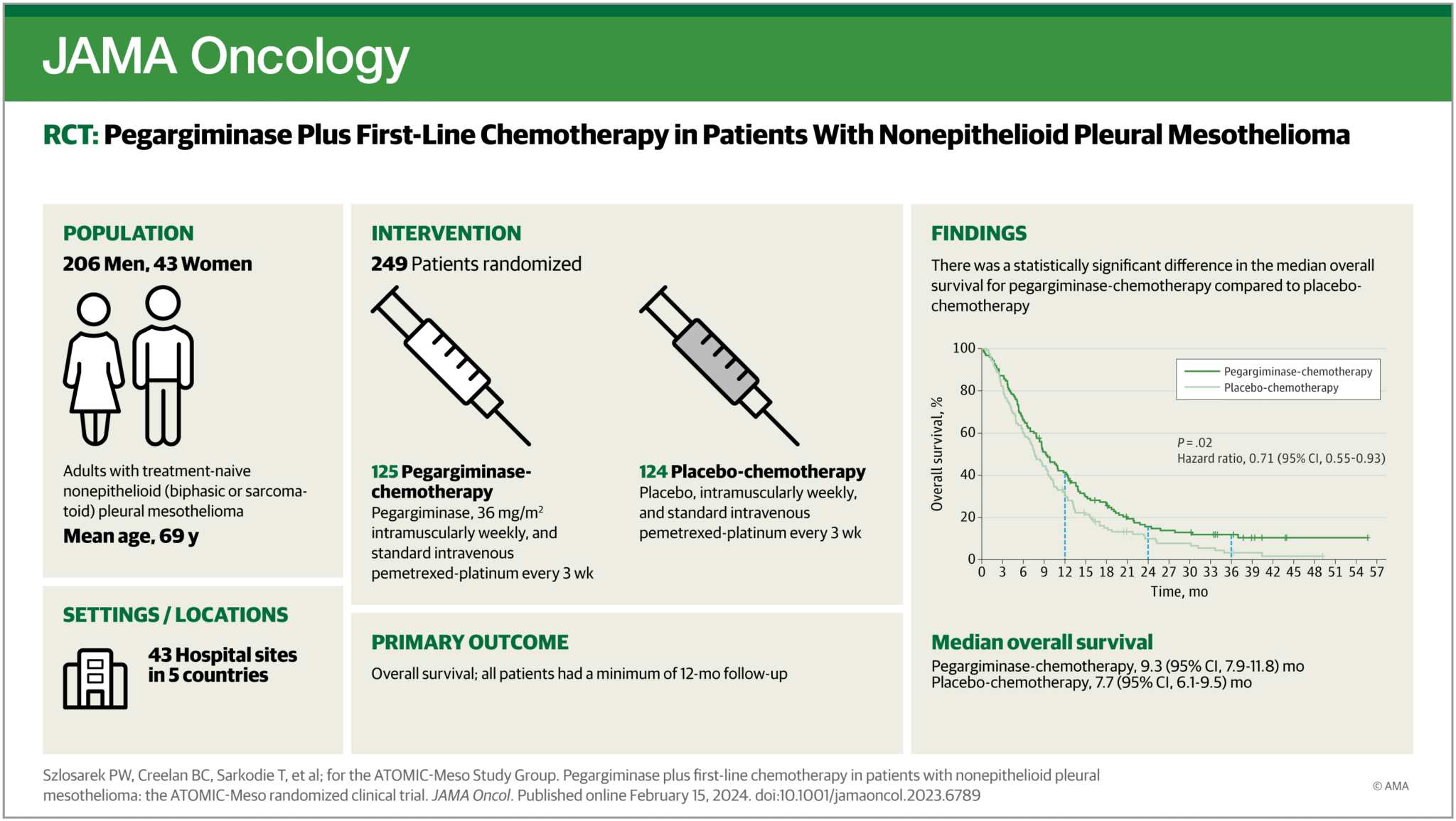
Data from a positive phase 2/3 clinical trial evaluating the effectiveness of a drug for mesothelioma was just published in the Journal of the American Medical Association (JAMA). The article titled Pegargiminase Plus First-Line Chemotherapy in Patients With Nonepithelioid Pleural Mesothelioma is available in the February 2024 issue.
This treatment, which is geared toward patients with sarcomatoid and biphasic mesothelioma who typically do not respond to standard chemotherapy treatment, works by depriving the cancer cells of arginine, an amino acid typically needed by the most aggressive mesothelioma tumors for proliferation. Deprivation of this amino acid works specifically in cancers with loss of a tumor suppressor enzyme, and sarcomatoid and biphasic mesotheliomas often fit the bill.
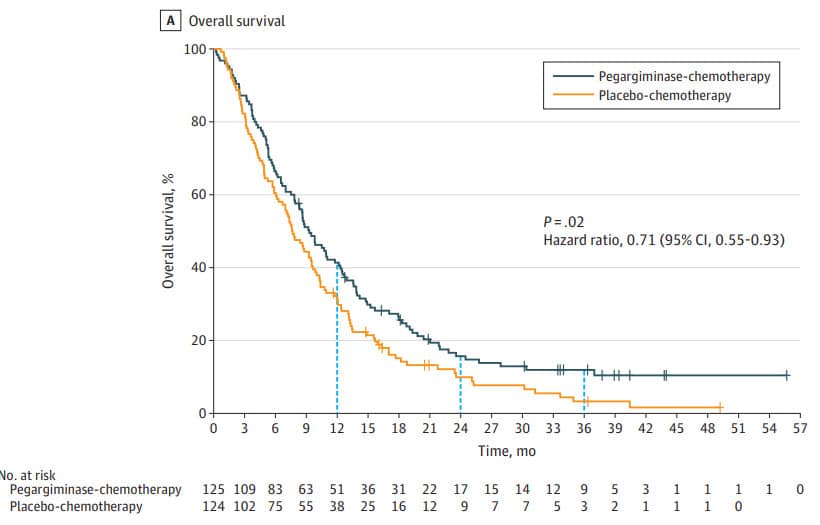
This clinical trial, called ATOMIC-Meso combined the arginine-depleting drug pegargiminase with standard chemotherapy (pemetrexed, also known as Alimta, and cisplatin). In the phase 3 of the trial, patients were randomized into two groups — one received chemotherapy plus pegargiminase, and the other received chemotherapy plus placebo. To be included in the study, patients were required to not have had any treatment, and to have nonepitheliod type of mesothelioma that is surgically nonresectable. The study ultimately included 249 patients. Overall survival improved significantly in patients receiving both chemotherapy and pegargiminase.
We spoke to Peter Szlosarek, MD, PhD, the principal investigator of this study and a medical oncologist and researcher at the Cancer Research UK Barts Cancer Institute in London, England. after this study’s data was released and he said the following:
“The top-line results from the ATOMIC-Meso study are nothing short of tremendous leveraging a novel area of cancer metabolism for patients by targeting arginine, specifically in non-epithelioid mesothelioma.”
“[These results] come after more than 5 decades since the first clinical use of asparaginase – the paradigm for amino acid depletion in cancer – namely childhood leukemia. ATOMIC-Meso has now set the bar for future studies of arginine deprivation in tumors requiring arginine as an essential amino acid on account of low expression of argininosuccinate synthetase 1 (ASS1),” he added.
He also indicated that because patients with sarcomatoid and biphasic mesothelioma tend to respond better to immunotherapy, further studies combining immunotherapy and pegargiminase are needed.
A previous article about this study was published at the time that data was first released in 2022: https://www.curemeso.org/2022/10/04/news-positive-results-reported-from-mesothelioma-phase-2-3-trial-for-sarcomatoid-biphasic-patients/.